|
|
International Journal of Arrhythmia 2015;16(1): 4-10.
|


Introduction
Electrophysiology study (EPS) and radiofrequency catheter
ablation (RFCA) are widely used for the diagnosis and treatment
of cardiac arrhythmias. EPS and RFCA are very complicated
procedures, requiring extended periods of time and advanced
technologies that vary according to the disease of the patients.
Fluoroscopy is used during EPS and RFCA to guide the catheter
through the vessels while viewing the fluoroscopy monitor to
reach the desired area to examine and monitor the procedure.
However, fluoroscopy exposes patients and staff members to
radiation, and this can result in possible skin damage, cancer, and genetic effects.1-3
An existing study on radiation exposure during RFCA showed
that the mean equivalent doses to the cardiologist’s left hand and
forehead were 0.24 mSv and 0.05 mSv, respectively, per RFCA
procedure, which was more than twice the mean dose for other
cardiology procedures.4 Another study reported that cardiac
electrophysiologists have high radiation exposure, with a median
of 4.3 mSv per year (range 3.5-6.1 mSv).5 In other studies of
RFCA, when the patients had an effective dose of 8.3 mSv for one
hour of fluoroscopy, they had a cancer risk of 480-650 per million
patients.6 Therefore, radiation exposure during electrophysiology
(EP) procedures is not insignificant for both patients and staff.
According to the European Committee on Radiation Risk,
when adults are exposed to a 10 mSv dose, 1 out of every 1,000
can be at risk for a possible solid tumor or leukemia in their
lifetime.7 Another report showed that if fluoroscopy exposure lasts
for more than 1 hour during an EP procedure, the dosage will
exceed the threshold and result in skin damage.8 However, most
cardiologists who perform procedures are not familiar with
radiation physics or methods to protect against it, or did not
received proper education about the risks of radiation, so
individual cardiologists are subjected to different levels of
exposure.9
Although many existing studies on radiation exposure in EP
procedures have focused on effective doses in patients,2,3,6,10 and
some have examined the radiation dose in both patients and staff
members,2,11,12 few have included cancer risk in their analysis.Therefore, this study aimed to determine the effective dose and
organ dose from radiation exposure during EP procedures in
patients, as well as to measure the risk of cancer from the effective
dose to staff members.
Materials and Methods
Patients
This study included 89 consecutive patients who received EP
procedures and cardiac implantable electronic device (CIED)
procedures from October 2011 to February 2012. All patients
gave informed consent. Three staff members (one cardiac
electrophysiologist, one radiologic technologist, and one nurse)
were included for the measurement of radiation exposure.
Radiation Dose Measurement
Procedures were performed using Philips Allura Xper FD20
fluoroscopy system (Philips Medical Systems, Eindhoven, The
Netherlands). The procedure was performed with fluoroscopy set
to “normal” and cinematic acquisition imaging frame rates set at
15 frames/sec in cardiac mode.
The tube voltage, tube current, and radiation exposure time
parameters were set at the time of installment with automatic
exposure control (AEC). Tube voltage, which was between
70-120 kV, was applied according to the type and size of the patient. A basic 1.5 mm Al and filtration of 0.2 mm Cu was
installed and a 0.1 mm Cu + 1.0 mm Al was applied for the
Selective Fluoro Prefilter.
Calculation of the Effective Dose and Organ Dose in Patients
The dose of radiation exposure to patients during the EP
procedure was measured by a dose area product (DAP) meter
(Diameter PTW, Freiburg, Germany), which was attached to the
collimator on the tube housing. The DAP value was used to
calculate the effective dose and organ dose with the PCXMC
Monte Carlo simulation program (version 1.5). The tube voltage
of the X-ray, tube current, and exposure time parameters were
performed by the AEC without a manual control. The tube
voltage of the AEC was flexible depending upon the size of the
patients and direction of the recording, ranging between 70-120
kV.
Radiation Exposure and Cancer Risk in Staff
In order to measure the radiation exposure to staff members, an
optically stimulated luminescent dosimeter (OSL) (Inlight/DSL
NanoDot Dosimeters, Landauer, Glenwood, IL. USA) was
attached to the protective equipment. The potential measurement
of the dose limit by the OSL was 100 ∂ Sv, the range of the energy
was 5 keV-20 MeV, and the accuracy was ± 5% of the standard
deviation. The OSL was attached at several locations: inside and outside of the gonad area, on the chest area of the apron, on the
lead goggles and thyroid protector of the operator, and also inside
and outside of the nurse's and radiological technologist’s apron.
After staff members had worn the OSL for 3 months, the data
collected from the OSL was sent to a specialist who determined
the radiation dose exposure at each site.
The effective dose of the staff members followed the Niklason
calculation,13 which is calculated as Deff = 0.02 (Hos-Hu) + Hu
(Hos is the dose outside of the lead apron and Hu is the dose
inside the lead apron). The lifetime attributable risk (LAR) of
cancer for the staff member was calculated based on the BEIR VII
study.14 That study showed the occurrence of cancer per every
100,000 people when they were exposed to 10 mGy annually
between the ages of 18 and 65; that data was directly applied to
the calculation of cancer risk to staff members. For example, when
the annual radiation exposure to staff members was 5 mSv, the
LAR was 5/10×3,059/100,000. This study measured the cancer
risk under the assumption that the staff members were
continuously exposed to radiation from the age of 18 to 65.
Results
Radiation Dose in the Patients
The average fluoroscopic duration was 20.8 minutes during the
procedure, the maximum was 68.4 minutes, and the total
fluoroscopic time was 1,040.1 minutes. The DAP value was an average of 112.0 Gy•cm2, and the maximum value was 519.6
Gy•cm2. Calculating the effective dose with the DAP value using
the PCXMC program resulted in an average of 35.9 mGy with a
maximum value of 166.5 mGy.
The average fluoroscopic time, DAP value, and effective dose
according to each EP procedure are shown in Table 1. The
fluoroscopic duration was the longest during AF ablation at 30.8
minutes, and the highest average effective dose in patients
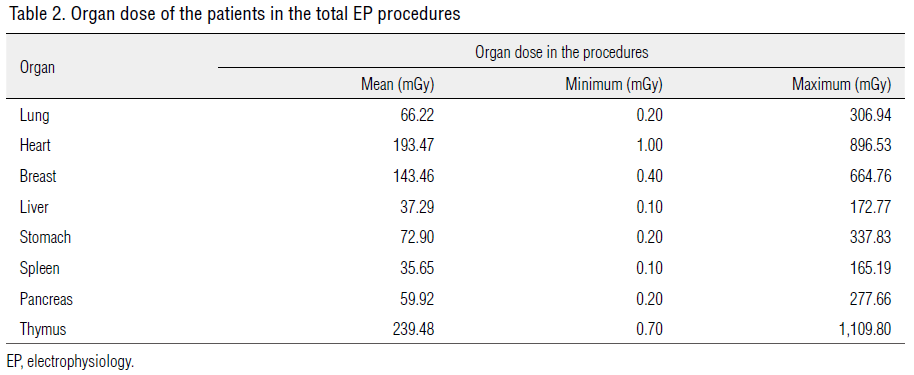
measured was 84.1 mGy. The organ dose converted with the
DAP value for the entire EP procedure in patients was highest in
the thymus, with an average of 239.48 mGy, followed by the heart
at 193.47 mGy, and breasts at 143.46 mGy (Table 2).

Effective Dose and Cancer Risk to Staff Members
The effective dose in staff members was calculated by reading
the OSL, which was worn for three months during the EP
procedures. The effective dose in the primary operator who was
closest to the patients for three months was 1.6 mSv, which
equates to an annual radiation exposure dose of 6.4 mSv. The
effective dose in the radiologic technologist was 0.98 mSv, and in
the nurse it was 0.75 mSv. The dose outside the apron for the
gonadal gland was 6,930 μSv, the dose in the area of the eyes was
3,200 μSv, and the thyroid was 4,020 μSv. These measurements
suggest that the outside of the apron in the gonadal gland area was
more exposed than the facial area. The attenuation rate, which
compared the readings inside and outside of the protective gear, was calculated at 83.8% for the apron of 0.5 mm thickness, 77.0%
for the 0.5 mm thyroid protector, and 50.4% for goggles with 0.07
mm lead thickness.
The cancer risk for male primary operators who are exposed to
6.4 mSv of radiation annually from the age of 18 to 65 is 1,958 per
100,000; in other words, 1 in every 51 operators would be at risk
for cancer. For operators consistently exposed to 6.4 mSv per year,
the mortality rate is 1 in every 92 operators (Table 3, 4).


Discussion
This study calculated the radiation dose in patients using the
DAP value in order to identify the radiation exposure dose to staff
members and patients during EP procedures while wearing
protective gear with an OSL attached during the procedure. The
amount of exposure of the patients as well as the risk of cancer was
also calculated. Interventions such as EPS procedures usually use
fluoroscopy. Because fluoroscopy is done by an AEC, there can be
difficulty in measuring the radiation exposure dose in patients.
Radiation can vary during fluoroscopy, and the exposed area of
the body constantly changes; therefore, in these kinds of
measurements, the dose-area product, DAP, is commonly applied.
The DAP value using the DAP meter is known as an effective way
to measure the amount of radiation in cardiac fluoroscopy and the
radiation area during fluoroscopy.15,16
In the previous studies, the DAP value during EPS procedures
was 11.6-251 Gy•cm2,4,17,18 and the effective dose in the patients was 17 mSv.4 Kovoor stated that it was 6.34 mSv for procedures
lasting 60 minutes,3 while Lickfett reported it was between 1.48-49.75 Gy•cm2.2
This study showed that the DAP value during EP procedures
was an average of 112.0 Gy•cm2 and the average effective dose in
the patients was 35.9 mSv, with a maximum of 166.5 mSv. This
study showed a higher average effective dose than previous studies.
During EP procedures, if patients are exposed to radiation for
longer than one hour, the threshold amount of radiation that is
critical for the skin will be reached, which has been reported
previously.8 During the AF ablation in this study, the exposure
time was an average of 30.8 minutes, and there was a long
exposure of 68 minutes, so the amount of radiation likely
exceeded the limit for skin damage.
The medical staff who perform electrophysiology procedures
often ignore or underestimate the danger of radiation. However,
the constant exposure to radiation during a few years of work or
life-long practice accumulates, and it can cause physical damage.
Furthermore, cardiologists are exposed to scattered rays, which
provide a fluctuating dose of radiation. In some cases, they are
exposed to direct rays. Because their hands, legs, and head area are
not properly protected, their accumulated dose can significantly
increase. In a study by Lucia Venneri,5 67% of the 5,164 cardiac
catheterization laboratory staff who worked with radiation in a hospital were exposed to radiation of 6 mSv or more. The study
also showed that staff members who worked at cardiology centers
might be exposed to the highest level of radiation. That study
noted that the annual radiation exposure for interventional
cardiologists averaged 3.3 mSv (2.0-19.6 mSv), and for
electrophysiology cardiologists it was 4.3 mSv (3.5-6.1 mSv),
equating to a fatal cancer risk of 1 in 384. The all-cause cancer risk
is 1 in 192. In the BEIR study,14 staff members exposed to 2 mSv
of radiation annually from the age of 18 to 65 had cancer risks of
612/100,000 for men or 859/100,000 for women. In other
words, the all-cause cancer risk for exposed staff was 1 in 136 and
the mortality rate was 1 in 245. Another study found that the
effective radiation dose in operators during percutaneous
coronary intervention procedures was 0.17-31.2 μSv and 0.24-9.6
μSv during EP and ablation procedures.19
In the present study, the effective radiation dose in staff
members during the EPS procedure was 1.6 mSv over three
months of exposure for primary operators, with an annual
exposure of 6.4 mSv. Extrapolating from this data, the all-cause
cancer incidence is 1 in 51, and the mortality rate is 1 in 92.
Therefore, although radiation exposure during EP procedures is
not immediately harmful in primary operators, over time, the
cumulative exposure can increase cancer risk. Radiation exposure
generally occurs due to scattered rays, except in the instances when operators put their hands into the fluoroscopic field to operate the
catheter. Fluoroscopy rays scatter in the iris of the radiation tube,
via leakage, and from reflection of patients.20 With the under tube
method, most rays reflected from the patients under the table,
which can directly affect the gonadal glands. In this study the
under tube method was used during EP procedures, and, as a
result, radiation exposure to primary operators appears higher in
the gonadal glands than in the eyes or thyroid gland. The results of
this study showed the same conclusion as the previous study.
Although operators wear aprons, lead shields, and goggles to
protect themselves, during long-term performance of these
procedures, it is impossible to avoid radiation exposure and its
effects. If operators fail to use protective gear or adjust the
exposure time properly, within a few years their eyes, skin, thyroid,
and gonadal glands may have increased cancer risk. The
attenuation rate of the protection equipment identified in this
study was 83.8% for a 0.5 mm lead apron, 77.9% for a 0.5 mm
lead thyroid protector, and 50.4% for 0.07 mm lead goggles. This
means that current equipment does not fully protect workers
from radiation exposure. The attenuation rate varied depending
on the kind. The most effective methods to reduce radiation
exposure during cardiac interventional procedures include
education regarding long-term exposure, developing a program to
decrease exposure to patients, installing proper equipment, and
using appropriate protective gear. Another method for reducing
exposure is in the operation of the machine. By setting a low level
for the fluoroscopy mode, preventing any unnecessary screen
widening, minimizing the source image distance (SID),
maximizing the source object distance (SOD), using a proper
filter, and selecting an image capture instead of spot image,
exposure to radiation for both patients and staff could be reduced.
The study had several limitations. First, the amount of indirect
radiation exposure in the patients could differ from the amount of
direct radiation exposure because of the calculation of the
radiation exposure using the PCXMC program with the DAP
value. Second, this study calculated the risk of cancer using the
BEIR VII study, but the potential cancer risk could be higher in
this study. This study ruled out many factors that could influence
the effective dose during the procedure, such as personnel,
mechanical, and environmental factors. Because the procedures
were performed with cardiac mode and a high frame rate setting, the amount of radiation exposure was higher than we expected.
Recently, the fluoroscopic setting was changed to “low” and
cinematic acquisition imaging frame rates changed to 3.75
frames/sec in cardiac EP mode. Therefore, we assume that the
amount of radiation exposure was much reduced as compared to
the study results.
References
- Park TH, Eichling JO, Schechtman KB, Bromberg BI, Smith JM, Lindsay BD. Risk of radiation induced skin injuries from arrhythmia ablation procedures.
Pacing Clin Electrophysiol.
1996;19:1363-1369.
- Lickfett L, Mahesh M, Vasamreddy C, Bradley D, Jayam V, Eldadah Z, Dickfeld T, Kearney D, Dalal D, Luderitz B, Berger R, Calkins H. Radiation exposure during catheter ablation of atrial fibrillation.
Circulation.
2004;110:3003-3010.
- Kovoor P, Ricciardello M, Collins L, Uther JB, Ross DL. Risk to patients from radiation associated with radiofrequency ablation for supraventricular tachycardia.
Circulation.
1998;98:1534-1540.
- McFadden SL, Mooney RB, Shepherd PH. X-ray dose and associated risks from radiofrequency catheter ablation procedures.
Br J Radiol.
2002;75:253-265.
- Venneri L, Rossi F, Botto N, Andreassi MG, Salcone N, Emad A, Lazzeri M, Gori C, Vano E, Picano E. Cancer risk from professional exposure in staff working in cardiac catheterization laboratory: Insights from the national research council's biological effects of ionizing radiation vii report.
Am Heart J.
2009;157:118-124.
- Perisinakis K, Damilakis J, Theocharopoulos N, Manios E, Vardas P, Gourtsoyiannis N. Accurate assessment of patient effective radiation dose and associated detriment risk from radiofrequency catheter ablation procedures.
Circulation.
2001;104:58-62.
- Wade JP. Estimation of effective dose in diagnostic radiology from entrance surface dose and dose-area product measurements.
Br J Radiol.
1998;71:994-995.
- Vano E, Arranz L, Sastre JM, Moro C, Ledo A, Garate MT, Minguez I. Dosimetric and radiation protection considerations based on some cases of patient skin injuries in interventional cardiology.
Br J Radiol.
1998;71:510-516.
- Rehani MM. Training of interventional cardiologists in radiation protection--the iaea's initiatives.
Int J Cardiol.
2007;114:256-260.
- Damilakis J, Theocharopoulos N, Perisinakis K, Manios E, Dimitriou P, Vardas P, Gourtsoyiannis N. Conceptus radiation dose and risk from cardiac catheter ablation procedures.
Circulation.
2001;104:893-897.
- Rosenthal LS, Mahesh M, Beck TJ, Saul JP, Miller JM, Kay N, Klein LS, Huang S, Gillette P, Prystowsky E, Carlson M, Berger RD, Lawrence JH, Yong P, Calkins H. Predictors of fluoroscopy time and estimated radiation exposure during radiofrequency catheter ablation procedures.
Am J Cardiol.
1998;82:451-458.
- Kadish AH, Mayuga KA, Yablon Z, Schaechter A, Goldberger JJ, Passman RS, Palmer A, Zimmer M, Davidson CJ. Effectiveness of shielding for patients during cardiac catheterization or electrophysiologic testing.
Am J Cardiol.
2001;88:1320-1323.
- Niklason LT, Marx MV, Chan HP. The estimation of occupational effective dose in diagnostic radiology with two dosimeters.
Health Phys.
1994;67:611-615.
- National Research Council (U.S.). Committee to Assess Health Risks from Exposure to Low Level of Ionizing Radiation. Health risks from exposure to low levels of ionizing radiation: Beir vii phase 2. Washington, D.C., National Academies Press.
2006.
- Le Heron JC. Estimation of effective dose to the patient during medical x-ray examinations from measurements of the dose-area product.
Phys Med Biol.
1992;37:2117-2126.
- Theocharopoulos N, Perisinakis K, Damilakis J, Varveris H, Gourtsoyiannis N. Comparison of four methods for assessing patient effective dose from radiological examinations.
Med Phys.
2002;29:2070-2079.
- Efstathopoulos EP, Katritsis DG, Kottou S, Kalivas N, Tzanalaridou E, Giazitzoglou E, Korovesis S, Faulkner K. Patient and staff radiation dosimetry during cardiac electrophysiology studies and catheter ablation procedures: A comprehensive analysis.
Europace.
2006;8:443-448.
- Davies AG, Cowen AR, Kengyelics SM, Moore J, Pepper C, Cowan C, Sivanathan MU. X-ray dose reduction in fluoroscopically guided electrophysiology procedures.
Pacing Clin Electrophysiol.
2006;29:262-271.
- Kim KP, Miller DL, Balter S, Kleinerman RA, Linet MS, Kwon D, Simon SL. Occupational radiation doses to operators performing cardiac catheterization procedures.
Health Phys.
2008;94:211-227.
- Tsapaki V, Kottou S, Vano E, Komppa T, Padovani R, Dowling A, Molfetas M, Neofotistou V. Occupational dose constraints in interventional cardiology procedures: The dimond approach.
Phys Med Biol.
2004;49:997-1005.
|
|
|
|