|
|
International Journal of Arrhythmia 2015;16(1): 19-24.
|


Introduction
Atrial fibrillation (AF) is a common cardiac arrhythmia
associated with a substantial risk of stroke and thromboembolism.1
Although the mechanism of stroke and thromboembolism in AF
patients is incompletely understood, the increased risk is mainly
due to the embolization of thrombus formed within the left atrial
appendage.2 There is increasing evidence that the increased risk of
stroke and thromboembolism in AF patients is facilitated by
increased thrombogenesis, with changes in the left atrial wall, a
prothrombotic or hypercoagulable state (including abnormalities
of hemostasis, thrombosis, and platelet and endothelial function),
and AF-related inflammation, leading to the fulfillment of
Virchow's triad.3-8
The renin-angiotensin-aldosterone system and inflammation
have been reported to affect the endothelial function,
prothrombotic and hypercoagulable state, and maintenance of
AF. It is not certain whether angiotensin converting enzyme
inhibitors (ACEIs) and statins improve the prothrombotic
condition in hypertension patients and reduce thromboembolism
in patients with persistent and permanent AF.
Material and Methods
All patients enrolled had hypertension, more than one known
risk factor for thromboembolism, and no prior history of taking
ACEIs, angiotensin receptor blockers, or statins. The enrolled
patients were randomly assigned to four groups. Group I (N=15;
M:F, 10:5; age, 49±6 years) received no ACEI or statin, group II
(N=17; M:F, 11:6; age, 48±6 years) received cilazapril 5 mg
without statin, group III (N=18; M:F, 9:9; age, 49±7 years)
received cilazapril 5 mg and atorvastatin 10 mg, and group IV
(N=16; M:F, 10:6; age, 45±3 years) received cilazapril 5 mg and
atorvastatin 40 mg.
Serum markers of endothelial function (von Willebrand factor
[vWF]), inflammation (quantitative and high sensitive C-reactive
protein [CRP], erythrocyte sedimentation rate [ESR]), and
coagulation (fibrinogen, fibrinogen degradation product [FDP],
d-dimer) were measured at baseline and 6 months. Blood samples
were obtained by atraumatic venipuncture in the morning, after
fasting for >12 hours. Blood was drawn without stasis into a tube
preloaded with trisodium citrate. vWF was measured by the enzyme-linked immunosorbent assay technique using commercial
reagents (R & D systems, MN, USA). CRP was measured by the
latex agglutination method (quantitative CRP, Behring
nephelometer analyzer; high-sensitivity CRP, Olympus AU
5400). Measurements of fibrinogen, FDP, and d-dimer were
performed with chromogenic assay (Sysmex CA1500, Sysmex
Corporation, Kobe, Japan).
All patients received antithrombotic therapy according to the
American College of Cardiology/American Heart Association
guideline. Drugs for heart rate control were prescribed equally
among all groups. Those who received anti-inflammatory drugs
such as steroids were excluded. Previously established and widely
accepted epidemiologic risk factors for thromboembolism
including old age (≥65 years), hypertension, diabetes mellitus,
heart failure, and history of embolism were investigated, and
echocardiographic parameters including left ventricular ejection
fraction (LVEF) and left atrial dimension were measured. Left
ventricular dysfunction was defined as LVEF less than 40%.
Exclusion criteria were recent (<6 months) myocardial infarction
or acute coronary syndrome, stroke, infection or inflammatory
disease, surgery, malignancy, thyrotoxicosis, and renal or liver
impairment.
Results are expressed as mean ± standard deviation.
Comparison of serum markers between groups were analyzed
using the paired t-test and analysis of variance. All statistical
calculations were performed using commercially available
statistical package software (SPSS version 12.0; IBM
Corporation, IL, USA). A P value <0.05 was considered
statistically significant.
Results
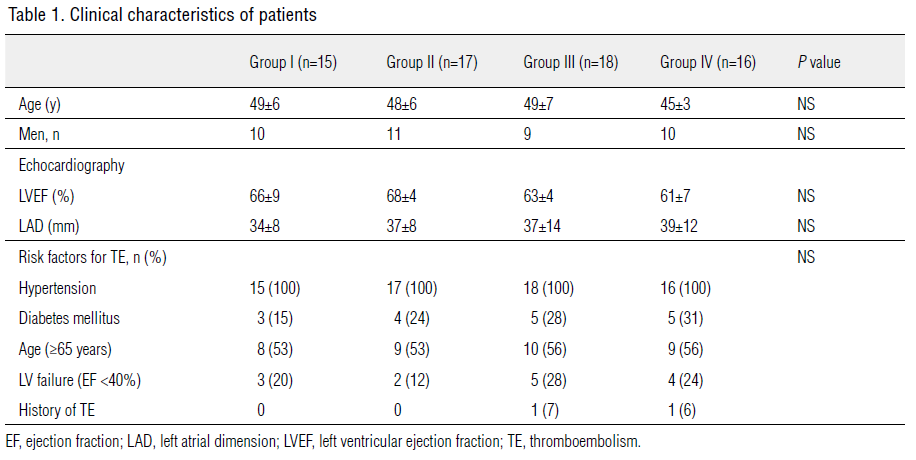
Clinical and demographic characteristics of the study
population are shown in Table 1. There were no significant
changes at 6 months or between-group differences in total
cholesterol, low-density lipoprotein cholesterol, high-density
lipoprotein cholesterol, and triglyceride levels at baseline and 6
months (Figure 1).
Markers of Endothelial Dysfunction
The vWF (factor 8-related antigen) level was significantly
decreased from baseline to 6 months in groups III and IV which
received statin (group III, 172% to 110%, p<0.001; group IV,
184% to 125%, p=0.021). There were no significant changes in
the vWF (ristocetin cofactor) level at 6 months or between-group
differences at baseline and 6 months (Figure 2).


Markers of Inflammatory Activity
There were no significant changes at 6 months or betweengroup
differences in ESR and CRP (quantitative and highsensitivity)
levels at baseline and 6 months (Figure 3).
Coagulation Markers
Fibrinogen levels were significantly decreased in group IV
(331±73 mg/dL vs. 275±57 mg/dL, p=0.047). However, there
were no significant changes in the other coagulation markers
(Figure 4).
Discussion
Endothelial dysfunction may lead to increased endothelial
adhesiveness to leukocytes and the production of procoagulant
and vasoactive molecules, cytokines, and growth factors.9 The
vWF is a multifunctional plasma protein that plays a very
important role in hemostasis following vascular injury. Circulating
platelets adhere to the injured site and initiate the process of
thromobosis, and subendothelial vWF mediates platelet adhesion
to the injured site. The vWF is secreted not only from the vascular
endothelium but also from the atrial endocardium in response to
vascular injury and vascular disease. Raised plasma levels of vWF
were reported to be associated with widespread endothelial
damage/dysfunction, atherothrombosis, left atrial endothelial
damage, and left atrial appendage thrombosis.10,11 Also,
thrombomodulin (TM), a transmembrane spanning protein
which can also be cleaved from the membrane to circulate in a
soluble form, is one of the major anticoagulant components of the
endothelial surface.12,13 Thrombin bound to TM consequently
loses its procoagulant and proinflammatory functions. It cannot
cleave fibrinogen or activate platelets and factor XIII.14 The
present study results suggest that markers of endothelial
dysfunction, especially the vWF, are associated with known
epidemiologic risk factors for thromboembolism in Korean AF
patients. Furthermore, ACEI and statin use could improve the
endothelial function after 6 months, regardless of the statin dose.
There is an apparent link between thrombogenesis and
inflammation. An established index of inflammation is
interleukin-6 (IL-6), which is a circulating cytokine produced by
monocytes, macrophages, T-lymphocytes, and endothelial cells.
IL-6 can induce a prothrombotic state by increasing the
expression of fibrinogen, tissue factor, factor VIII, and von
Willebrand factor, as well as by activating endothelial cells and
increasing platelet production.3-5 Elevated CRP levels have been
reported in AF patients, reflecting an inflammatory state, which
could promote the persistence of AF.15 Although the CRP level
was increased and correlated with some known risk factors for
thromboembolism in previous studies, the use of ACEI or statin
did not affect these markers or composite endpoint even with
high doses. These results suggested the lesser role of inflammation
in terms of thromboembolic risks in Korean AF patients.
The level of hemostatic activation may also reflect the
underlying mechanism of thromboembolism, especially
pronounced in cardioembolic stroke.16 Fibrin D-dimer assay is
based on the production of cross-linked fibrin by thrombin,
making it a sensitive marker of fibrin turnover, and allows the
recognition of activated coagulation.17 Coagulation markers were
substantially increased and associated with some risk factors for
thromboembolism, and the use of high-dose atorvastatin at 40 mg
could reduce fibrinogen levels after 6 months. Further wellcontrolled
studies are required to evaluate the clinical effect of this
dose and treatment duration.
There were no remarkable lipid profile changes even after 6
months of treatment with high-dose statin therapy. This result
may have been associated with patient compliance. The
mechanism of the pleiotropic effect of statins which involves an
improvement in endothelial function and coagulation is unclear.
While the benefits of antithrombotic therapy in preventing
stroke in AF patients are being increasingly recognized, further
developments in thromboprophylaxis are needed, especially as
warfarin confers the inconvenience of regular monitoring of prothrombin time (PT) and the benefits of aspirin are
inconsistent. Current clinical practice for prevention of
thromboembolic stroke in AF patients is limited not only by the
low efficacy of antiplatelet therapy but also by the hemorrhagic
complications and the inherent need for PT monitoring with
warfarin therapy.18
In conclusion, the present study showed some beneficial effects
of ACEI and high-dose statin on endothelial function and
coagulation. The use of these drugs in addition to the
conventional treatment with anticoagulation drugs may be useful
for the prevention of thromboembolic events in high-risk patients
or for secondary prevention of thromboembolism. The present
study findings should be verified in further randomized
controlled studies.
References
- Wolf PA, Abbot RD, Kannel WB. Atrial fibrillation as an independent risk for stroke: the Framingham Study.
Stroke.
1991;22:983-988.
- Hart RG, Halperin JL. Atrial fibrillation and stroke: concepts and controversies.
Stroke.
2001;32:803-808.
- Lip GY, Lowe GD, Rumley A, Dunn FG. Increased markers of thrombogenesis of chronic atrial fibrillation: effects of warfarin therapy.
Br Heart J.
1995;73:527-533.
- Lip GY. Does atrial fibrillation confer a hypercoagulable state?
Lancet.
1995;346:1313-1314.
- Kumagai K, Fukunami M, Ohmori M, Kitabatake A, Kamada T, Hoki N. Increased cardiovascular clotting in patients with chronic atrial fibrillation.
J Am Coll Cardiol.
1990;16:377-380.
- McEver RP. Adhesive interactions of leukocytes, platelets, and the vessel wall during hemostasis and inflammation.
Thromb Haemost.
2001;86:746-756.
- Kerr R, Stirling D, Ludlam CA. Interleukin 6 and haemostasis.
Br J Hematol.
2001;115:3-12.
- Joseph L, Fink LM, Hauer-Hensen M. Cytokines in coagulation and thrombosis: a preclinical and clinical review.
Blood Coagul Fibrinol.
2002;13:105-116.
- Blann AD. Endothelial cell activation, injury, damage and dysfunction: separate entities or mutual terms.
Blood Coagul Fibrinolysis.
2000;11:623-630.
- Conway DSG, Pearce LA, Chin BSP, Hart RG, Lip GY. Prognostic value of plasma von Willebrand factor and soluble P-selectin as indices of endothelial damage and platelet activation in 994 patients with nonvalvular atrial fibrillation.
Circulation.
2003;107:3141-3145.
- Roldan V,Marin F, Garcia-Herola A, Lip GY. Correlation of plasma von Willebrand fator levels, an index of endothelial damage/dysfunction, with two point-based stroke risk stratification scores in atrial fibrillation.
Thromb Res.
2005;116:321-325.
- Califano F, Giovanniello T, Pantone P, Campana E, Parlapiano F. Clinical importance of thrombomodulin serum levels.
Eur Rec Med Pharmacol Sci.
2000;4:59-66.
- Maruyama I. Thrombomodulin, an endothelial anticoagulant; its structure, function and expression.
Jpn Circ J.
1992;56:187-191.
- Fuentes-Prior P, Iwanaga Y, Huber R, Pagila R, Rumennik G, Seto M, et al. Structural basis for the anticoagulant activity of the thrombin-thrombomodulin complex.
Nature.
2000;404:518-525.
- Chung MK, Martin DO, Sprecher D, Wazni O, Kanderian A, Carnes CA, et al. C-reactive protein elevation in patients with atrial arrhythmias. Inflammatory mechanisms and persistence of atrial fibrillation.
Circulation.
2001;104:2886-2891.
- Feinberg WM, Erickson LP, Bruck D, Kittelson J. Hemostatic markers in acute ischemic stroke. Association with stroke type, severity, and outcome.
Stroke.
1996;27:1296-1300.
- Lip GY, Lowe GD. Fibrin D-dimer turnover;a useful clinical marker of thrombogenesis?
Clin Sci.
1995;89:205-214.
- Levine MN, Raskob G, Landefeld S, Kearon C: Hemorrhagic complications of anticoagulant treatment.
Chest.
2001;119:108S-121S.
|
|
|
|