Ambulatory Hemodynamic Monitoring of Pulmonary Hypertension
Article information
Abstract
Elevated pulmonary artery pressure, typically called “pulmonary hypertension (PH)”, may occur owing to several pathophysiological conditions. The World Health Organization (WHO) proposed 5 classifications of PH, primarily based on causality, in order to guide research and provide a framework for therapeutic intervention. The WHO group II PH is most common and characterized by a mean pulmonary artery (PA) pressure higher than 25 mmHg that is associated with a pulmonary capillary wedge pressure greater than 15 mmHg. This hemodynamic profile suggests that PA pressure is elevated in cases of left ventricular (LV) failure, such as severe mitral valve disease (stenosis or regurgitation), LV diastolic failure, or LV systolic failure. Patients with elevated PA pressure experience abnormal dyspnea on exertion as well as severe symptoms such as dyspnea at rest. Further elevations in PA pressures can cause fluid to exudate fluid from the intravascular to the extravascular space, leading to pulmonary edema and severe, potentially life-threatening decompensation that require in-hospital care. Recently developed ambulatory implantable hemodynamic monitors help physicians remotely measure direct PA pressure from patients’ homes. Monitoring PA pressures allows healthcare specialists to monitor patient stability by evaluating the response to therapy adjustments, ultimately reducing hospitalization.
Background
Blood circulation in the cardiovascular system relies on pressure gradients introduced by contractions of the heart. The right ventricle (RV) pumps blood through the vasculature of the pulmonary circuit to the lungs for oxygenation and then to the left atrium (LA). Blood moves from the LA to the LV, which pumps blood through the vasculature of the systemic circuit to the rest of the body, and then back to the right atrium (RA) and RV. The systolic and diastolic phases of the cardiac cycle induce maximum and minimum pressures, respectively, that supply the hemodynamic driving force for circulation.
Pulmonary artery pressures are usually assessed by using right heart catheterization techniques in which a fluid-filled catheter is advanced through a vein into the RA, RV, and pulmonary artery (PA). Most fluid-filled catheters have an inflatable balloon at the tip that can serve to wedge the catheter into a vessel, which essentially creates a conduit from the PA to the LA. The so-called PA wedge pressure (i.e., occlusion pressure) is a right-sided approach to measure LA pressure (LAP). The fluid-filled catheter is connected to a properly positioned transducer that measures the pressure pulses on a diaphragm in the transducer. This information is then recorded by a special hemodynamic package in the catheterization laboratory. Several calculations can then be made to estimate vascular resistance and intravascular volume, and to classify pulmonary hypertension (PH). These calculations further classify World Health Organization (WHO) group II PH as “pre-capillary,” arising from vasoconstriction; “post-capillary” or “passive,” arising from excessive intravascular volume; or “mixed,” arising from vascular remodeling, which result in increased pulmonary capillary resistance and increased intravascular volume (Table 1) [1]. Therapeutic interventions designed to alleviate symptoms arising from WHO group II PH should target specific etiology, which illustrates the importance of understanding which component is particularly prominent in the PH pathophysiology.

Clinical classification of pulmonary hypertension, as proposed at the Fourth World Symposium on Pulmonary Hypertension in 2008.
The focus of this review is the monitoring of PH heart disease, particularly with the use of implantable hemodynamic monitors (IHMs).
Pulmonary Hypertension Management
Rationale for Pharmacological Therapy
Left-sided heart failure (HF), the most common cause of WHO group II PH, can arise from several different mechanisms, including the following: weakened myocardial contraction resulting in reduced fraction of blood ejected during the cardiac cycle (HF with reduced ejection fraction [EF], HFrEF), stiffened myocardium with reduced filling compliance resulting in elevated LV end-diastolic pressures reflected backline into the LA and pulmonary venous system (HF with preserved EF, HFpEF), mitral valvular disease resulting in a significant amount of blood ejection backline into the LA and pulmonary veins (regurgitant fraction), or mitral stenosis resulting in marked elevation of LAP due to fused mitral valve leaflets and resistance to forward flow. Regardless of the underlying myocardial or valvular problem, the resulting pathological condition is elevated pulmonary venous and arterial pressures. Patients with WHO group II PH have worse mortality and morbidity outcomes.
Therapeutic interventions generally focus on treating the underlying left-sided HF with the goals of improvement of myocardial performance, prevention of worsening of the underlying disease, and repair or replacement of the mitral valve when appropriate. Consensus exists for the treatment of HFrEF, which is calculated as a percentage according to the following formula:
Cardiac function is often quantified by the volume of blood pumped per minute, known as cardiac output (CO), which is a product of stroke volume (SV) and heart rate (HR). Treatment of HFrEF is focused on two adverse reactions that occur in response to reduced CO. The first reaction is considered “neurohormonal” and involves the response of the central nervous system to baroreceptor input from the periphery. With a sensed decrease in CO, the brain assumes that the body is dehydrated or hemorrhaged, which are both associated with reduced blood volume. Therefore, central reflex output involves activation of the renin/angiotensin/aldosterone system that causes peripheral vasoconstriction and encourages sodium/water retention in the nephron. In addition, parasympathetic nervous control of the heart withdraws in concert with sympathetic activation, leading to short-term increases in contractility and CO. Angiotensin II and aldosterone both maintain the mean arterial pressure (MAP) by increasing total peripheral resistance (TPR) to blood flow. The homeostatic mechanisms in play are specifically designed to acutely maintain MAP to ensure appropriate organ perfusion. The determinants of MAP are as follows:
As can be seen from this formula, sympathetic nervous activation can increase SV and HR, while angiotensin II and aldosterone can alter TPR in order to maintain adequate organ perfusion with preservation of MAP.
Patients with chronic HF, however, have a constant signal to the brain that CO is depressed, leading to constantly elevated circulating neurohormone levels. The long-term effects of this situation are quite different from those in short-term dehydration or hemorrhage. Angiotensin II and aldosterone stimulate fibroblast activity that leads to fibrosis, which further reduces the strength of myocardial contraction and causes “adverse remodeling” of the myocardium and vasculature. Chronically elevated sympathetic activity accentuates the effects of angiotensin II, independently stimulates fibroblasts and inflammatory markers, and leads to electrophysiological abnormalities conducive to sudden cardiac death from malignant tachyarrhythmias.
Pharmacological therapies for HF patients, then, focus on each of these known abnormalities by inhibiting the production of angiotensin II from angiotensin I by using angiotensin-converting enzyme inhibitors or by blocking the angiotensin receptor by using angiotensin receptor blockers. Mineralocorticoid receptor antagonists (MRA) also are used to attenuate the ill effects of aldosterone activation. Beta-blockers attenuate the effect of chronically elevated sympathetic activation in patients with HF. Use of these pharmacological agents increases survival rates, improves quality of life, decreases the need for hospitalizations, and reverses adverse structural remodeling.
The second major area of therapy for patients with HFrEF that also applies to those with HFpEF is control of excessive intravascular volume, which commonly occurs despite the use of neurohormonal antagonists. Loop diuretic therapy is the mainstay of volume control. These agents exert their effect at the loop of Henle in the nephron by inhibiting sodium reabsorption and increasing the volume of urine produced. Many patients who require chronic loop diuretic therapy become resistant to the effects primarily because of development of hypertrophy of the distal convoluted tubule, which can also retain sodium and water. Combinations of loop and distally active diuretics should be administered multiple times to remove excess intravascular volume, and restore stable symptoms and prevent disease progression. Arginine vasopressin antagonists, a third diuretic class that inhibit the formation of aquaporins in the collecting duct of the kidney, recently became available. This results in free-water release into the urine. These drugs are approved in the United States for the treatment of dilutional hyponatremia but are commonly used worldwide as adjunctive diuretic therapy for patients with HF.
No consensus exists about the treatment of patients with HFpEF, except to control systemic hypertension and avoid excessive intravascular volume. This is because most clinical trials that test specific strategies have not been successful in changing clinical outcomes in this group of patients. The TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist) trial examined the effect of chronic MRA use in patients with HFpEF. Although the primary end point of mortality difference was negative, hospitalization rates were lower in the treatment group than in the placebo group [2]. Therefore, the only clinical pharmacological intervention to possibly have an effect in patients with HFpEF is MRA using spironolactone.
Rationale for Device Therapy
Despite significant reductions in overall mortality rates and progression of disease through neurohormonal intervention, the incidence of malignant, potentially lethal arrhythmias remains high, especially in those with HFrEF (LVEF < 35%). Clinical trials that intend to address the unacceptably high rate of sudden arrhythmic death were developed especially after implantable cardioverter defibrillators (ICDs) became smaller and easier to implant. Mortality reductions were consistently observed across a wide range of patients with HFrEF initially described as LVEF < 30% and associated with coronary artery disease but subsequently broadened to include patients with LVEF < 35% due to any etiology. Subsequently, it became clear that ICDs, once implanted, simply monitor for potentially lethal arrhythmias with the intention of early termination to prevent sudden death. This device therapy may increase hospitalizations and has no effect on HF disease progression. Therefore, this clarifies that the “value” of ICDs would be enhanced if the physiological information that devices monitor could be harvested as a clinical marker of HF disease status.
The first of such an evaluation came with measurement of HR variability (HRV) by using the atrial-to-atrial depolarization interval sensed by the atrial lead of a dual chamber device. These data demonstrated that patients with very low HRV (i.e., low parasympathetic control and activated sympathetic tone) were at high risk of decompensation and hospitalization. In addition, it was clear that HRV declined as patient clinical status declined. This monitoring feature only allowed assessment of status, but could not remotely guide therapy intervention. Other features of HF pathophysiology were also monitored, including daytime and nighttime HR, activity level as sensed by the accelerometer system of the device, along with atrial and ventricular arrhythmias.
The next device class, atrial-sequential biventricular pacing, was developed to specifically treat interventricular and intraventricular conduction delay in patients with symptomatic HFrEF. Electrical conduction abnormality, particularly left bundle branch block with widened QRS complex on resting electrocardiography, is known to promote disease progression by producing intraventricular and interventricular mechanical dyssynchrony. Application of biventricular pacing, also known as cardiac resynchronization therapy (CRT), in the treatment of electrical/mechanical dyssynchrony resulted in reduced need for hospitalizations, reversal of adverse ventricular remodeling, and improved survival.
Monitoring features initially associated with CRT devices included measurement of intrathoracic impedance as a marker of excessive volume in patients with chronic HF. Many of these monitoring features provided clinical utility in remote assessment of patients with HF and determining changes in their pathophysiology. However, they lacked the ability to guide clinical decision making to control pulmonary pressure, which is the key factor responsible for worsening of clinical status and increasing the need for hospitalization. Therefore, this clarifies that a more direct measurement of PA pressure or volume associated with acute increases in pulmonary pressures may improve outcomes in patients with secondary PH.
Current Pulmonary Hypertension Monitoring
A successful ambulatory monitoring strategy should take into account the patient’s cardiovascular health and risk status, while closely monitoring the patient’s response to treatment and avoiding overtreatment. Medications should be continually updated according to the changing cardiovascular landscape of each patient. Consequently, regular monitoring is essential and can be used to detect particularly dangerous levels of hypertension before symptoms worsen and hospitalization is required.
Current ambulatory IHMs rely on either (1) thoracic impedance for estimates of downstream fluid volume overload (i.e., congestion) or (2) pressure for direct intracardiac or intravascular hemodynamic measurements.
Impedance-based IHM
With more than 300,000 ICD systems implanted each year [3], extending the same circuitry to monitor related cardiovascular diseases has an obvious appeal. The accumulation of fluid that leads to elevated PA pressure, electrically conductive by nature, reduces the electrical impedance across the thoracic cavity. Low amplitude current pulses can be injected from the RV lead electrodes to the ICD or CRT device case, and intrathoracic impedance can be measured based on Ohm’s law. In clinical studies, impedance-based monitoring has proven successful for the identification of thoracic fluid congestion and prediction of congestive HF events that require hospitalization. Because of these studies, thoracic impedance monitoring has been recommended by the Heart Rhythm Society for management of chronic HF [4].
Impedance-based IHMs have been released by several medical device manufacturers as diagnostic features in ICDs. These IHM implementations include Medtronic OptiVol®, St. Jude Medical CorVue™, and Biotronik Thoracic Impedance. All three systems periodically monitor intrathoracic impedance and compile daily values. The impedance trend can be compared to patient-specific references to provide the clinician with the magnitude and duration of any deviations. When detected early, these congestive events can be addressed therapeutically prior to the exacerbation of HF symptoms and further progression of hypertension.
The Medtronic OptiVol® Fluid Status Monitoring feature is available in Medtronic devices equipped with defibrillation leads. The intrathoracic impedance is estimated from the RV coil electrode of the defibrillation lead to the device case. Sudden and sustained drops in impedance, relative the patient’s average, initiate a notification of a congestive event. Left untreated, such notifications have been associated with higher risks of rehospitalization in a series of clinical trials (PARTNERS-HF [Program to Access and Review Trending Information and Evaluate Correlation to Symptoms in Patients With Heart Failure], OFISSER [OptiVol Fluid-Index InSync Sentry Registry], FAST [Fluid Accumulation Status Trial], and CONNECT [Clinical Evaluation of Remote Notification to Reduce Time to Clinical Decision]) [5] and higher risks of mortality in a retrospective analysis of remote monitoring data [6]. Consistent with the indirect nature of impedance-based assessments of hemodynamics, OptiVol® demonstrated a congestive event detection sensitivity of 20.7% in the SENSE-HF (Sensitivity of the InSync Sentry feature for the Prediction of Heart Failure) trial [7]. Unfortunately, a prospective randomized clinical trial that tested if threshold crossings tied to a patient alert system would improve outcomes in patients with HF found that this system actually increased healthcare utilization and hospitalizations when compared to usual care.
The Biotronik Thoracic Impedance feature is available in Biotronik devices with defibrillation leads. Thoracic Impedance uses an approach similar to OptiVol® but measures impedance from the distal electrode of the RV lead to the device case. Results from the clinical trial have not been reported yet.
The St. Jude Medical CorVue™ Congestion Monitoring feature, unlike Medtronic and Biotronik, estimates intrathoracic impedance with a multiple vector approach, incorporating a RV ring to the device case and a RV coil to case circuits. The DEFEAT-PE (Detect Fluid Early From Intra-thoracic Impedance Monitoring) clinical trial showed a CorVue™ sensitivity of 21.6% in order to maintain a false-positive rate of 0.9 per patient-year [8].
All methods of intrathoracic impedance have a potential to provide earlier detection of volume accumulation in patients with HF. This notification service is likely helpful in triaging follow-up but may not be useful in remote adjustment of medical therapies, such as intensification of diuretics or other HF medications. Furthermore, these markers were validated as a surrogate for PA pressures. Recently developed technology now allows the direct measurement of pulmonary pressures by using IHMs, which not only provides early detection of fluid accumulation but also allows monitoring of medication effect to determine when pressure returns to baseline. This process represents a significant breakthrough in complete and personalized disease management for patients with WHO group II PH.
Pressure-based IHMs
To date, multiple IHMs based on intracardiac or intravascular pressure sensors have been released, including the following: ImPressure/RemonCHF (Boston Scientific, Natick, MA), Chronicle (Medtronic, Minneapolis, MN), HeartPOD™/Promote® LAP (St. Jude Medical, Sylmar, CA), and the CardioMEMS™ HF System (St. Jude Medical, Sylmar, CA). Most of these systems were tested in clinical trials that included patients with secondary PH and symptomatic HF, either HFpEF or HFrEF, with the intention to guide medical therapy and maintain stability, rather than react to decompensation. This approach was hypothesized to be superior to standard tools in terms of assessing and maintaining HF patients, with the goal of reducing the need for hospitalization. Several sensor systems have been developed to measure PA pressures, of which some have been tested in clinical trials. The common features of these systems are that they are implantable and can remotely transmit information about PA pressures from the patients’ home environment.
The ImPressure/RemonCHF system includes a battery-powered pressure sensor and transducer housed in a titanium case, and is fixed in the PA by a stent-like nitinol wire. PA pressure signals can be wirelessly downloaded from the implant by a handheld unit by using ultrasound, enabling remote monitoring. The safety and feasibility of the device were demonstrated by the PAPIRUS (Pulmonary Artery Pressure by Implantable device Responding to Ultrasonic Signal) II clinical study, which revealed no serious adverse events and accuracy relative to a high fidelity pressure catheter over 6 months [9]. The clinical benefit of this PA pressure monitor is currently being evaluated in the PAPIRUS III trial.
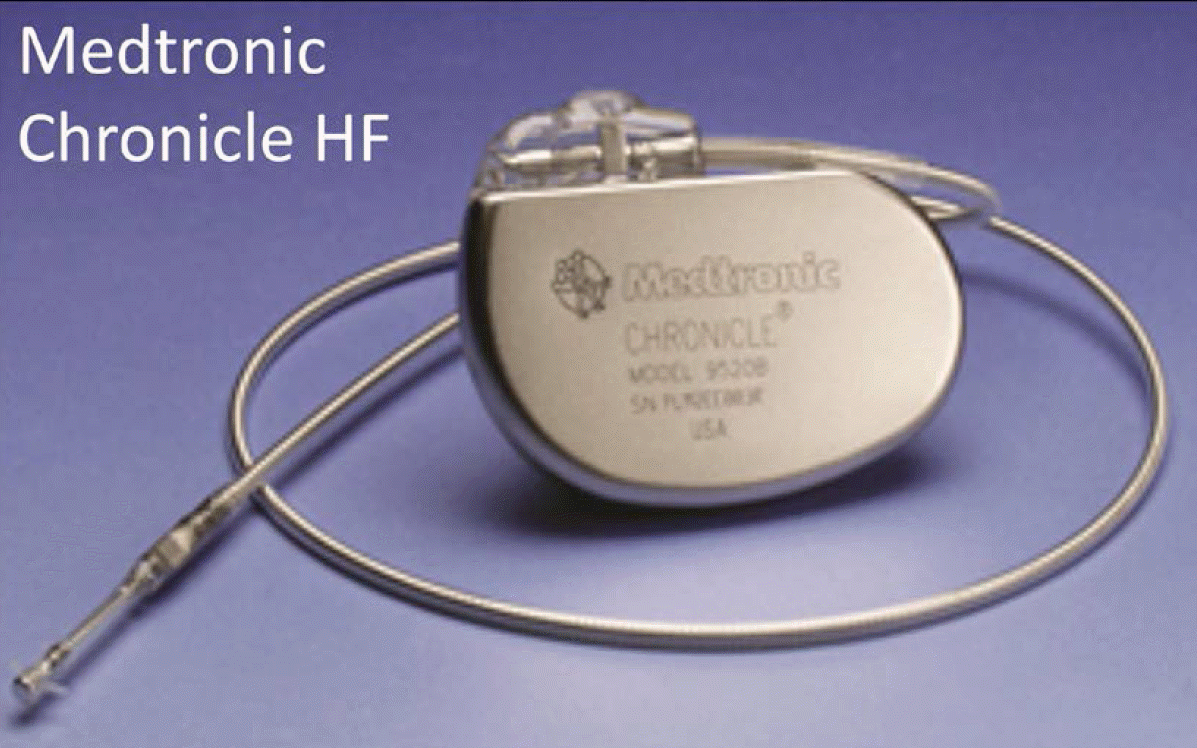
The Chronicle system shares an overall design and delivery technique to the traditional pacemaker, and has a pectoral case connected to a tined RV lead equipped with an intraventricular pressure sensor (Figure 1). The pressure-sensitive capacitor in the sensor measures RV Ps, Pd, and PP, which can be used to estimate PA Pd. In addition, the lead tip electrode provides electrograms and HR that are used to detect bradyarrhythmias and tachyarrhythmias. These metrics, including body temperature and activity level, are continuously monitored and stored in device memory. Data can be wirelessly downloaded to external transmission units and sent to clinicians for potential adjustments to treatment. The accuracy of Chronicle pressures, relative to measurements via right heart catheterization, has been demonstrated in several small-scale studies [10-12]. In addition, the device was successfully used to quantify the short- and long-term effects of multiple hypertension medications [13,14]. In terms of safety, the COMPASS-HF study demonstrated negligible sensor failures and device-related complications [15]. In terms of clinical efficacy, the device showed a 21% reduction in HF-related hospitalization, which is greater than that by optimal medical therapy alone [15].
The HeartPOD™ system directly measures LAP with a pressure sensor-tipped lead that is transseptally introduced from the RA into the LA, and fixed with nitinol anchors at the septum. Like Chronicle, this system was also modeled after the traditional pacemaker. The lead is connected to a pectoral case that includes an antenna coil for wireless radiofrequency transmissions to an external handheld unit. The HOMEOSTASIS (Hemodynamically Guided Home Self-Therapy in Severe Heart Failure Patients) trial showed low rates of device failure (5% at 2 yr and 12% at 4 yr) and strong concordance with the standard LAP estimation technique, which measures pulmonary capillary wedge pressure by using a Swan-Ganz catheter [16]. Long-term efficacy is currently being studied in the LAPTOP-HF (Left Atrial Pressure Monitoring to Optimize Heart Failure Therapy) trial. The HeartPOD™ system has also been incorporated into St. Jude Medical ICDs, labeled as the Promote® LAP system. While the progression of hypertension and HF can be observed earlier by monitoring left heart pressures rather than right heart pressures and thoracic impedance, left heart interventions may add to the risk of stroke.
The CardioMEMS™ HF System includes an implanted sensor, delivery system, and external transmission unit (Figure 2). The sensor is a hermetically sealed microelectromechanical system (MEMS) comprised of an inductor coil and pressure-sensitive capacitor. Right heart catheterization is used to deliver and calibrate the sensor at a distal pulmonary artery. Nitinol wire loops extending from the sensor expand upon deployment to ensure PA fixation. The sensor does not rely on leads, batteries, or active-fixation mechanisms. The resonant frequency of this passive circuit is pressure dependent. The external transmission unit powers the sensor wirelessly by using RF energy and converts frequency to pressure. PA pressure waveforms, namely Ps, Pd, MAP, and HR, are wirelessly transmitted from the home or hospital to a secure database for clinician assessment. CardioMEMS™, like Chronicle, provides right-side heart pressures. However, the PA Pd waveform that is provided by both devices can be used to estimate downstream LA pressures [17]. In terms of safety, the CHAMPION (CardioMEMS Heart Sensor Allows Monitoring of Pressure to Improve Outcomes in NYHA Class III Patients) trial demonstrated negligible sensor failures and device-related complications, similar to Chronicle [18]. In terms of clinical efficacy, the device showed a 39% reduction in HF-related hospitalization at 15 months, which is greater than that by optimal medical therapy alone [18].
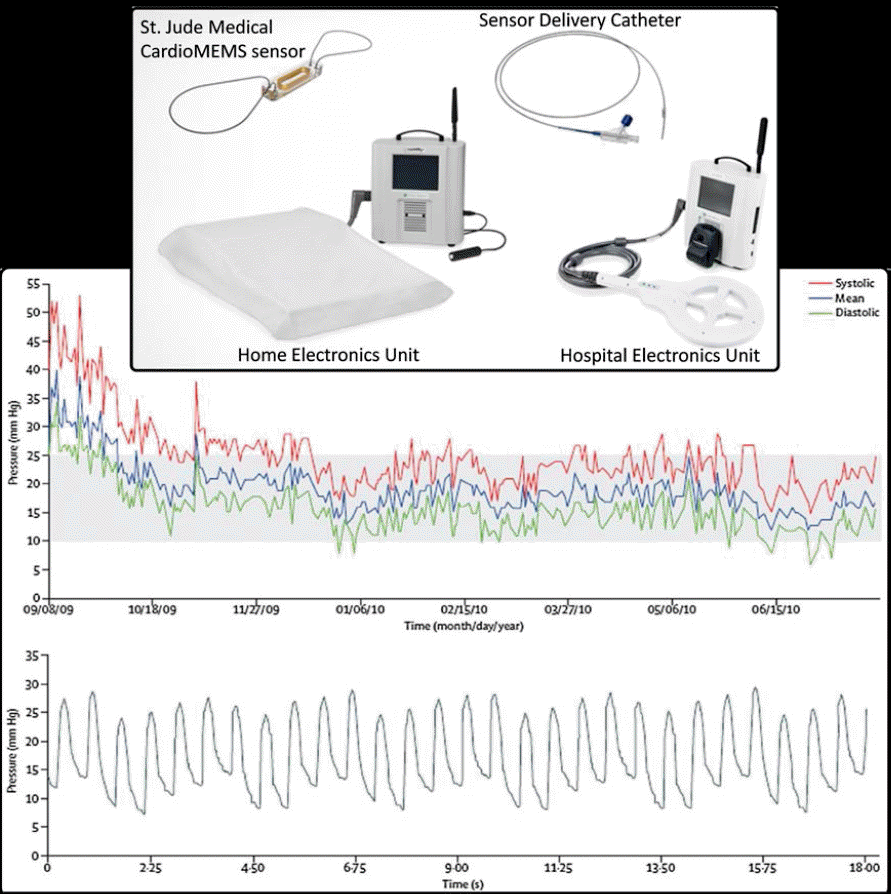
The St. Jude Medical CardioMEMS™ HF System. Individual components are shown at the top, and examples of long-term pressure trends and an individual pressure waveform are provided at the bottom.
Patients with confirmed secondary PH also benefitted from remote monitoring of PA pressures in the CHAMPION trial [19]. Patients with PH were at higher risk of adverse events and mortality, but treatment group outcomes were better than those in the control group during the entire follow-up of the trial. Patients with HFpEF represented a predetermined subgroup analysis in the CHAMPION Trial. Medication adjustments included diuretics and addition of vasodilator therapy in this group, with the expressed purpose of reducing PA pressures and avoiding excursions from baseline values. This strategy reduced HF hospitalizations by 50% in the treatment group, a higher reduction rate than that in the control group [20], representing one of the first successful treatment strategies for this group of patients.
Future Considerations
Ambulatory approaches for the long-term monitoring of progression of hypertensive heart disease have moved in the direction of IHMs. The unique ability of IHMs to report daily trends of pressure or congestion allow physicians to respond early to the warning signs of disease progression. Treating the underlying condition with adjustments to medical therapy can prevent disease progression and deterioration of artery disease, HF, arrhythmias, renal disease, and stroke. With the increase in the global prevalence of hypertension and related conditions, however, healthcare professionals are in dire need of aggressive advances in monitoring and management.
Patients with hypertension are often indicated for ICD and CRT devices, hence the popularity of impedance-based congestion monitoring. The cardiovascular health of these patients relies on optimal functioning of the implanted device to maximize the efficiency of the heart as a hemodynamic pump. Systematic optimization of atrial and ventricular timing can be costly and time consuming, typically involving echocardiography for hemodynamic assessment, and is often avoided entirely. Furthermore, the optimal parameter set identified at implant has been shown to alter with time and may require reoptimization. By using an IHM as a surrogate for echocardiography to evaluate ICD or CRT efficacy could streamline the process of tailoring therapy to each patient. Moreover, the development of closed loop IHM-ICD or IHM-CRT communication may enable frequent, automated device-based reoptimization of electrical therapy. This form of automated reprogramming can address changes in physiological conditions, such as posture or physical activity.
Patient compliance is a major limiting factor of the success of hypertension management. The IHM feedback provided to physicians for adjustments in therapy often involve limited communication and participation on the patient side. The evolution and popularity of wearable health monitors synchronized to smartphones is indicative of the growing interest in patients’ control of their own well-being. Patients may be able to log meals, exercise, and sleep times that can be linked to IHM data, which may give their physicians a more complete hemodynamic report. Drawing patients closer to their physician-therapy loop, with easily accessible and understandable updates of their progress, may improve treatment compliance and outcomes.
The accuracy of impedance-based monitoring in detecting congestion at discrete time points has been demonstrated. Nonetheless, the dynamics of the pulmonary and systemic vasculatures are complex. In practice, reducing the entire cardiovascular system to a gross electrical impedance measurement is an oversimplification with limited specificity for detecting serious hypertensive episodes that require medical intervention. The growing financial burden of unnecessary hospitalization has placed an emphasis on avoiding false-positive episode detections, which has been achieved at the expense of reduced sensitivity. For example, most approved thoracic impedance algorithms require about 2 weeks of continuous fluid congestion to signal a congestive event. Advances in the sensitivity and specificity of instrument that detecting fatal congestive events will allow earlier presymptomatic hospitalization and only when necessary, thus reducing overall healthcare cost.
Summary
Pulmonary hypertension is most often associated with left HF and involves a downward spiral of RV strain and failure. Although available medications are proven to improve survival in patients with left HF, changes in volume status causes shortness of breath and may require hospitalization. Long-term, out-of-clinic monitoring has been introduced to help physicians remotely tailor the combination and dosage of medications to fit each patient’s pulmonary pressure profile. In addition, remote monitoring of pulmonary pressures or congestion can warn the physician and patient of impending decompensation episodes before symptoms worsen and if hospitalization is required. The development of ambulatory implantable hemodynamic monitoring systems may reduce hospitalization rates. Although an increase in both the prevalence and cost of HF are expected to continue in the future, there will advancement in medical technology that targets early diagnosis and treatment of elevated pulmonary pressures.